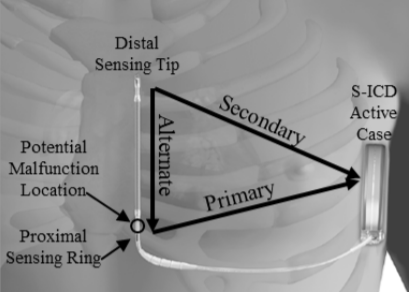
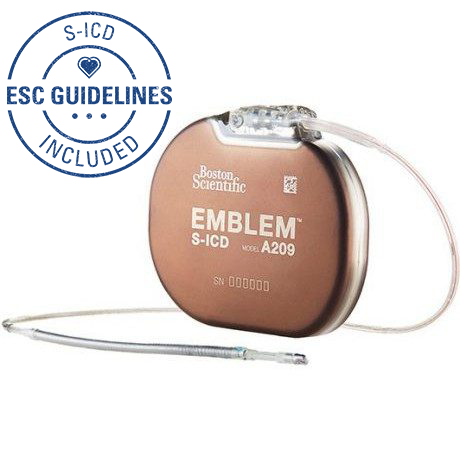
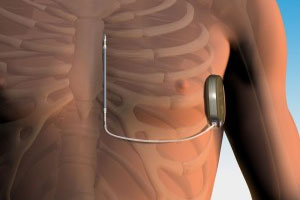
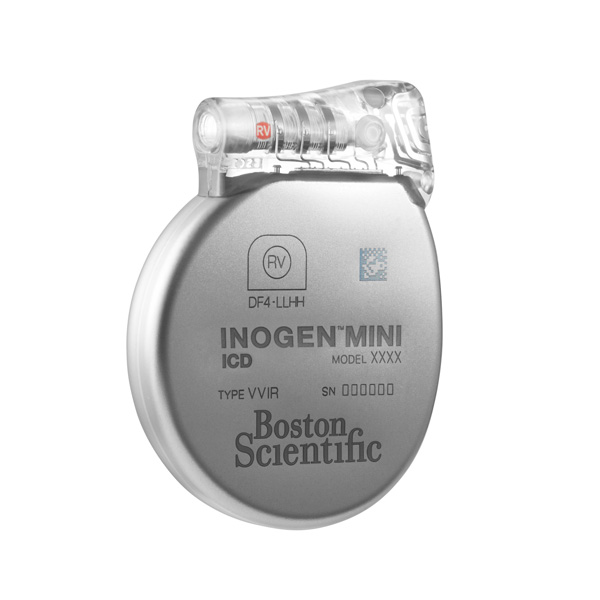
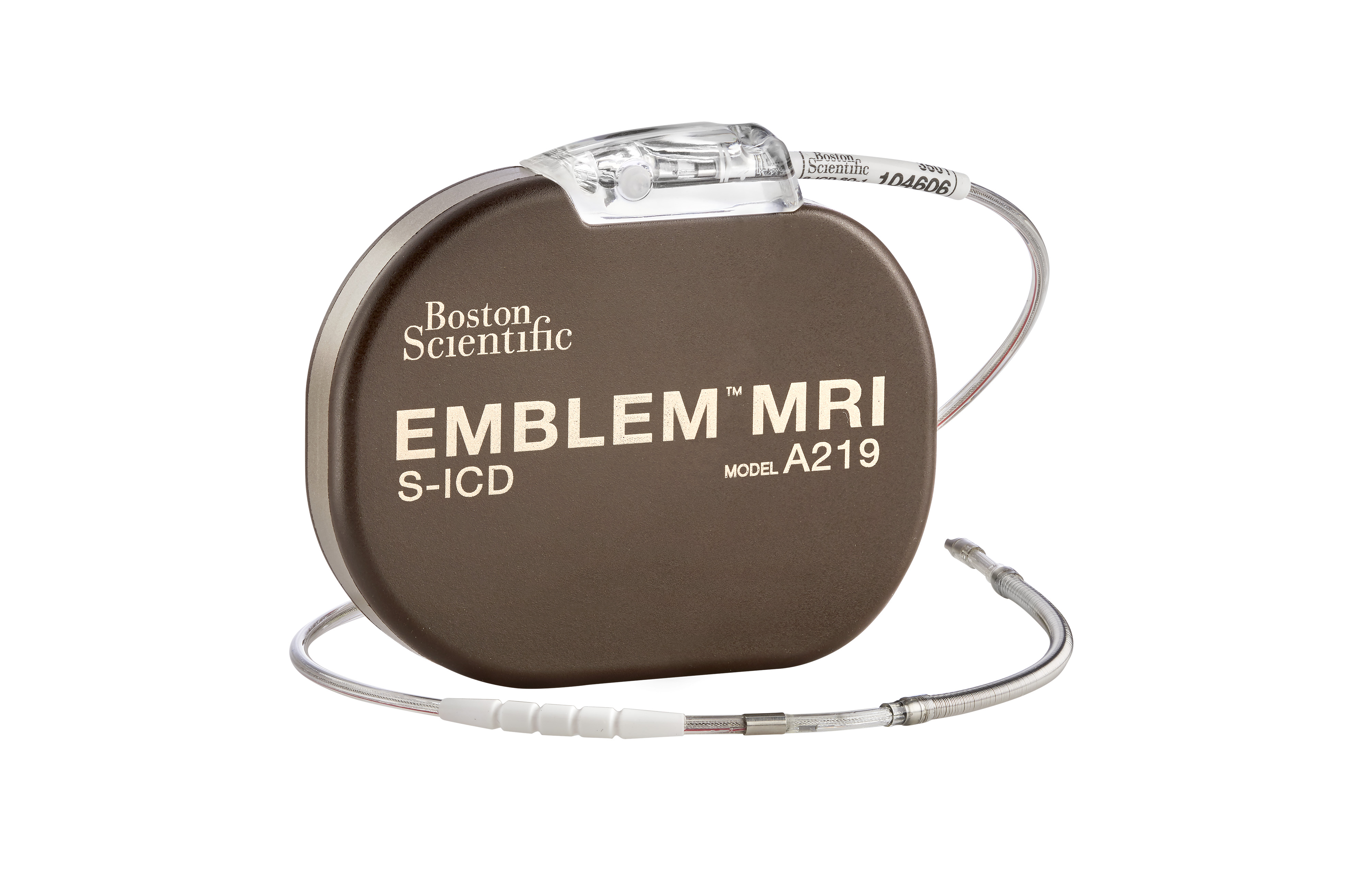
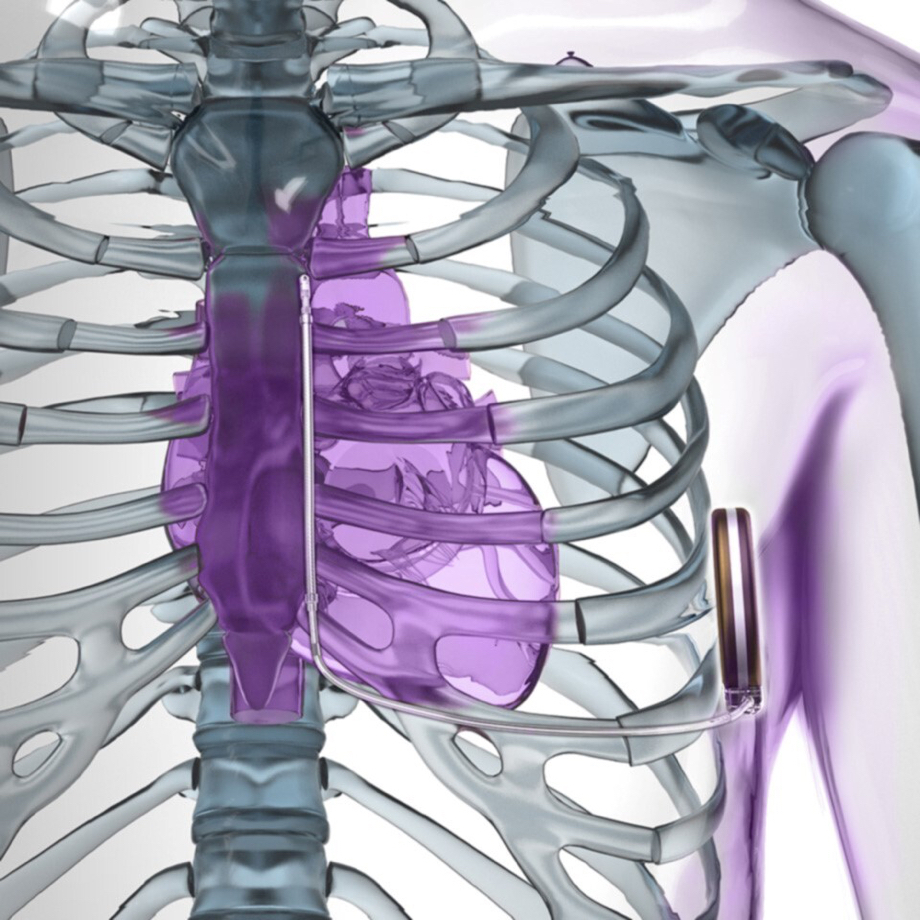
What patients should know about the EMBLEM™ Subcutaneous Implantable Defibrillator (S-ICD) System advisories
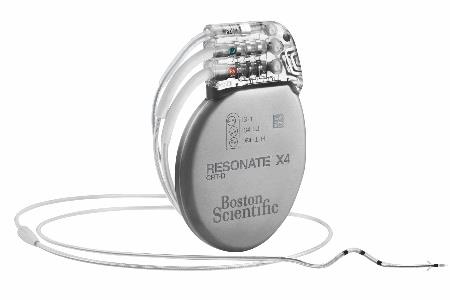
Boston Scientific Announces US FDA Approval For MRI Labeling On High-Voltage Devices And US Launch Of Resonate Devices with The HeartLogic Heart Failure Diagnostic

Boston Scientific announces scheduled presentations at Heart Rhythm Society 2017 - Cardiac Rhythm News