Cyanide attack at low-spin iron(II)–diimine complexes: the structure of a cyanide-containing derivative of a leaving ligand

Spin Crossover in Tetranuclear Cyanide-Bridged Iron(II) Square Complexes: A Theoretical Study | Inorganic Chemistry
Mixed Ligand Complexes of Iron(II) and (III) with Cyanide and Aromatic Di-imines | Journal of the American Chemical Society
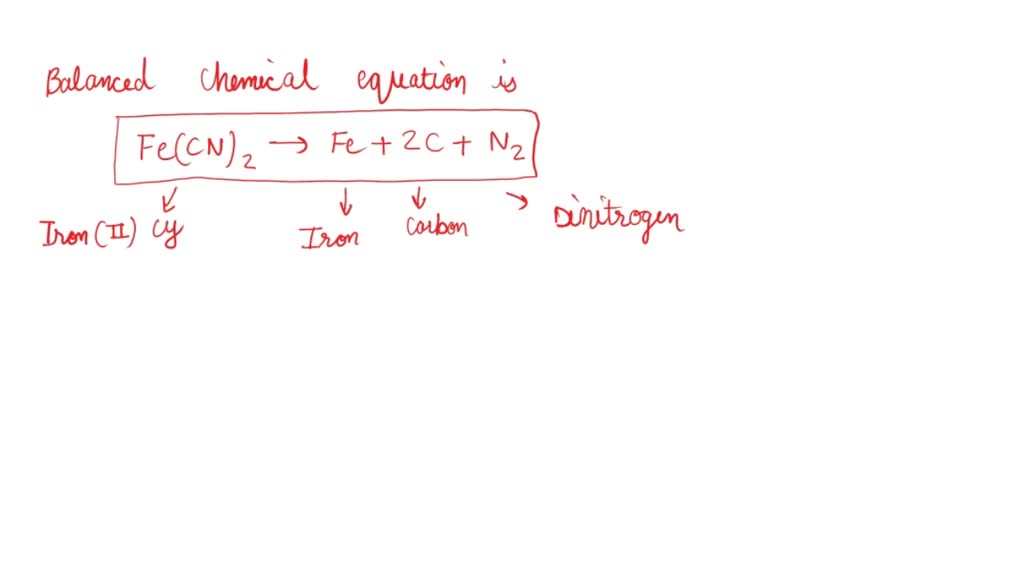
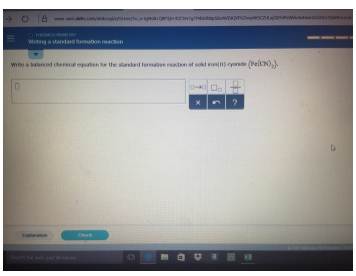
SOLVED: Write a balanced chemical equation for the standard formation reaction of solid iron(II) cyanide (Fe(CN)):

Temperature and salt effects of the kinetic reactions of substituted 2-pyridylmethylene-8-quinolyl iron (II) complexes as antimicrobial, anti-cancer, and antioxidant agents with cyanide ions
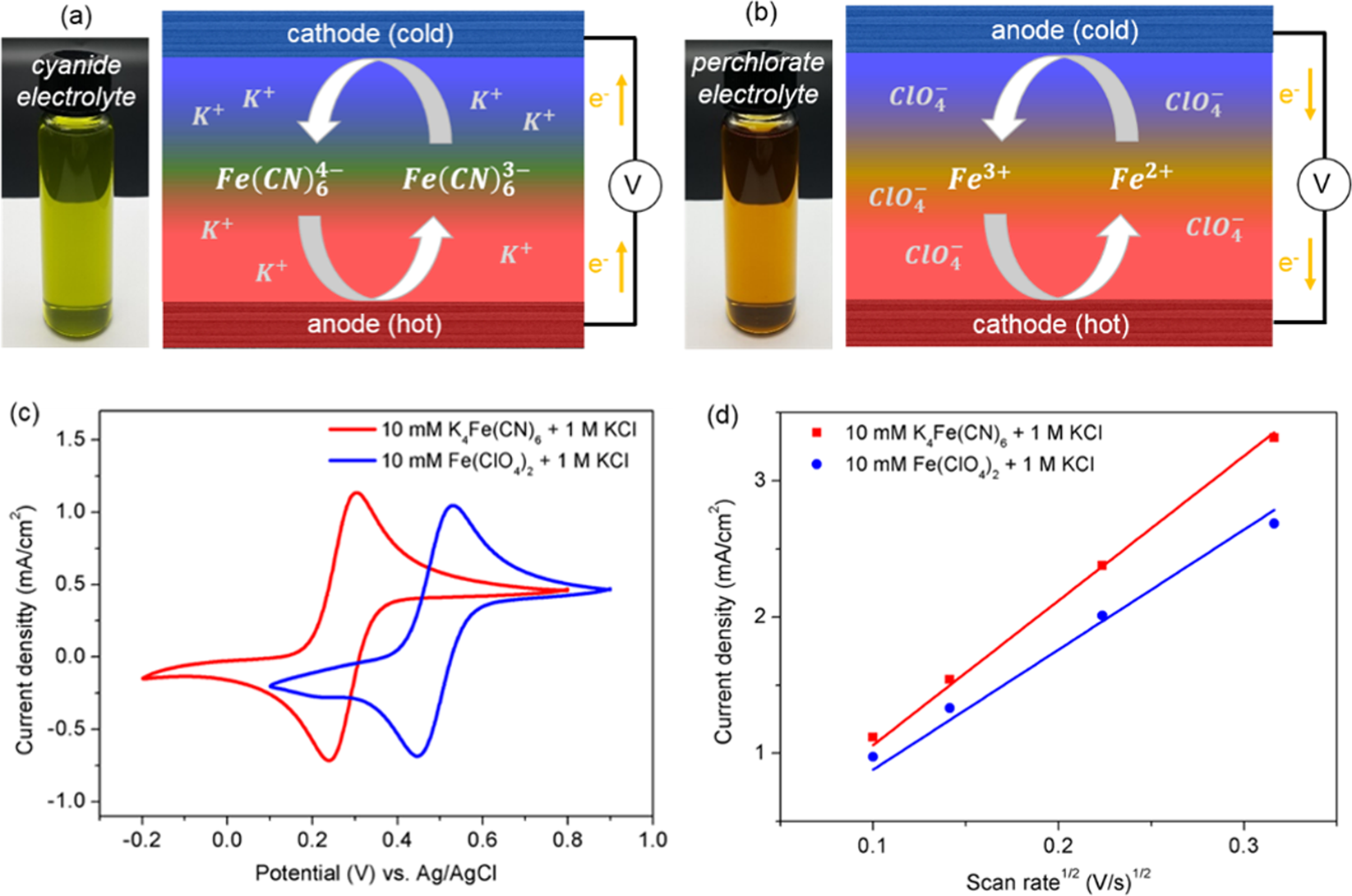
Iron (II/III) perchlorate electrolytes for electrochemically harvesting low-grade thermal energy | Scientific Reports

Comparison of Cyanide and Carbon Monoxide as Ligands in Iron(II) Porphyrinates - Li - 2009 - Angewandte Chemie International Edition - Wiley Online Library


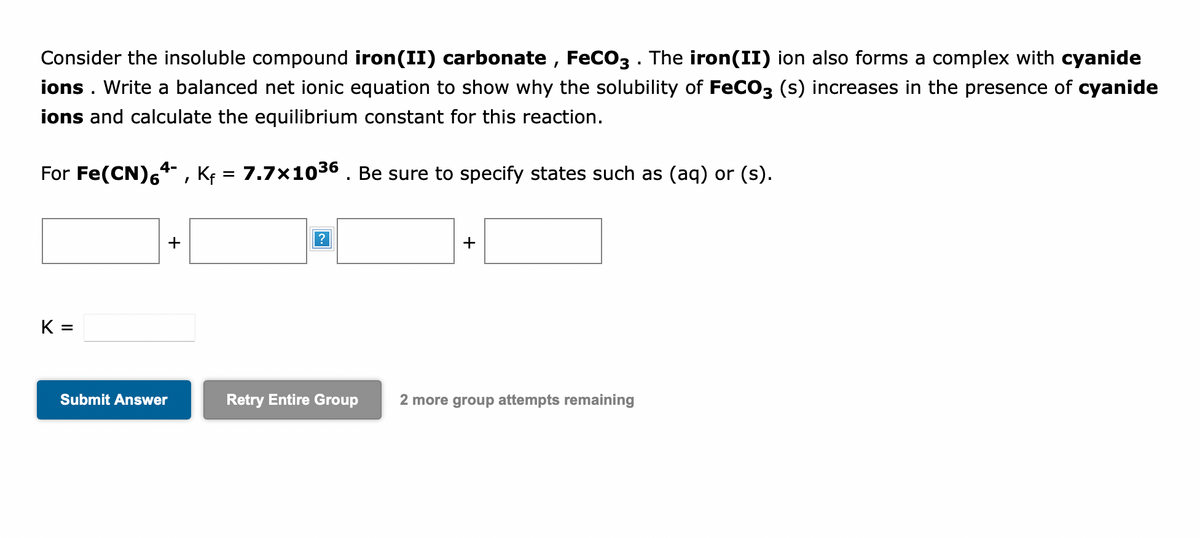
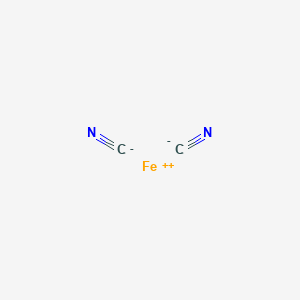



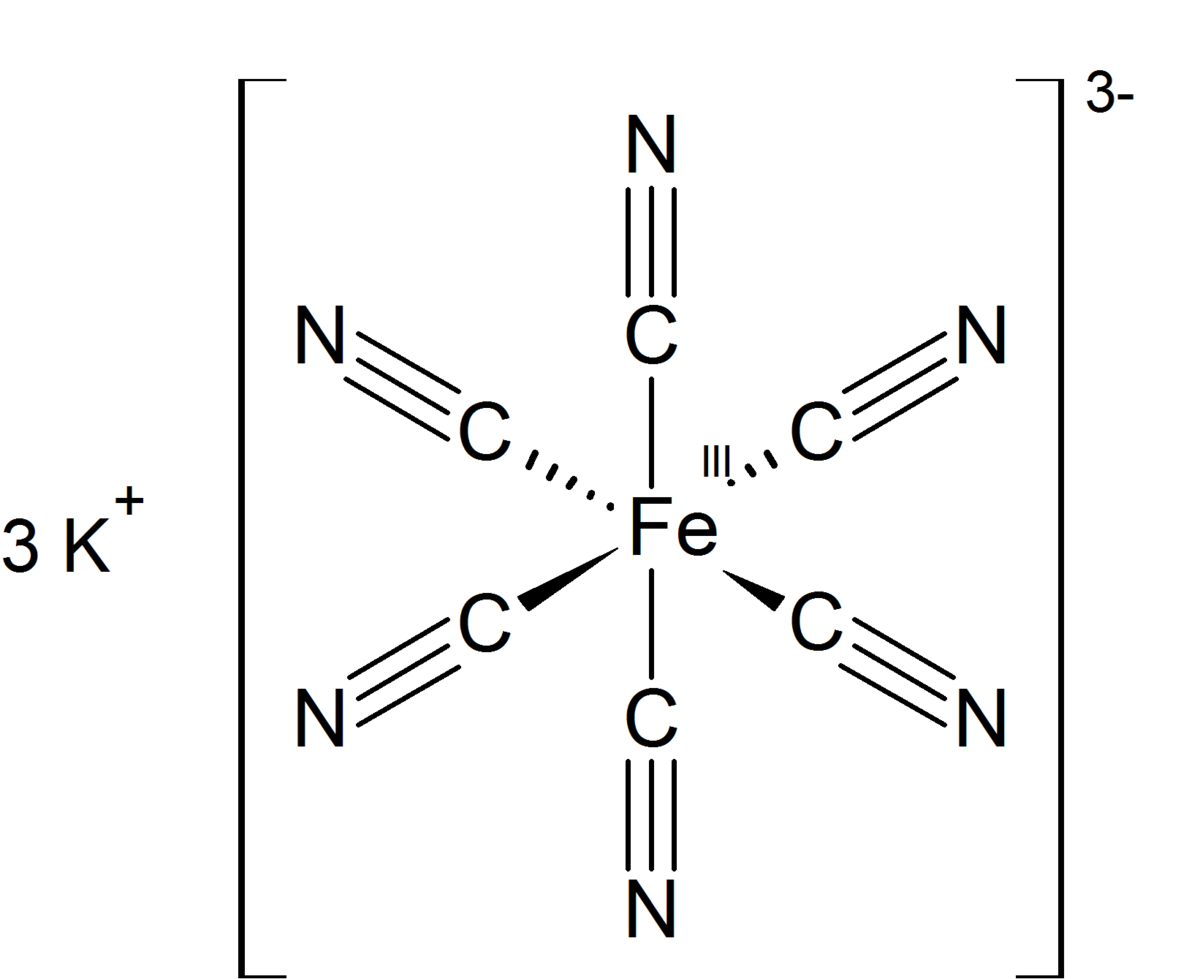




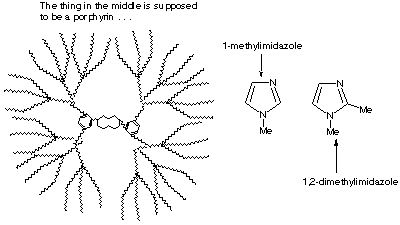



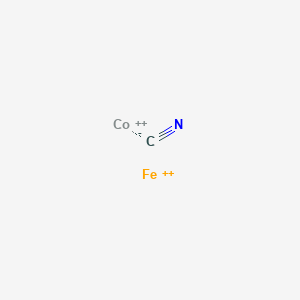

![PDF] Cyanide Ligand Assembly by Carbon Atom Transfer to an Iron Nitride. | Semantic Scholar PDF] Cyanide Ligand Assembly by Carbon Atom Transfer to an Iron Nitride. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/0e69e374e4e33906723bcb4c8a2f906521dcdb48/3-Figure1-1.png)