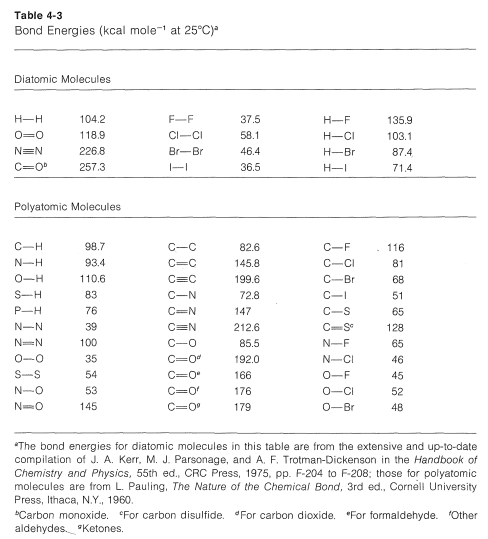
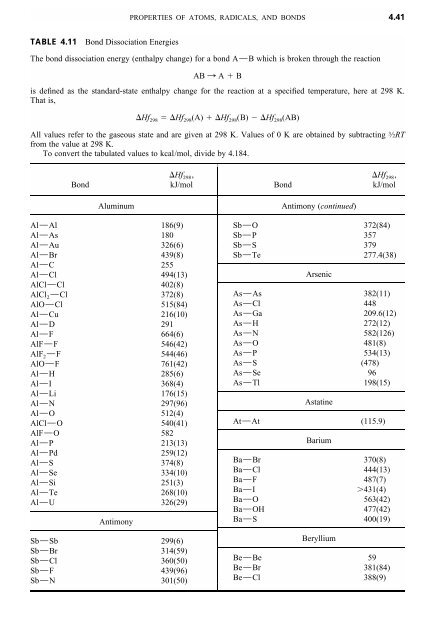
WebElements Periodic Table » Periodicity » Bond enthalpy of diatomic M-B molecules » Periodic table gallery
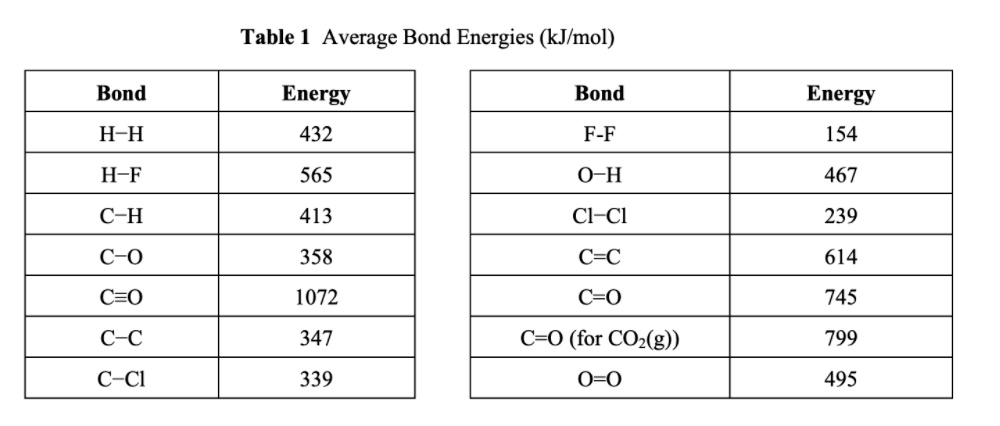
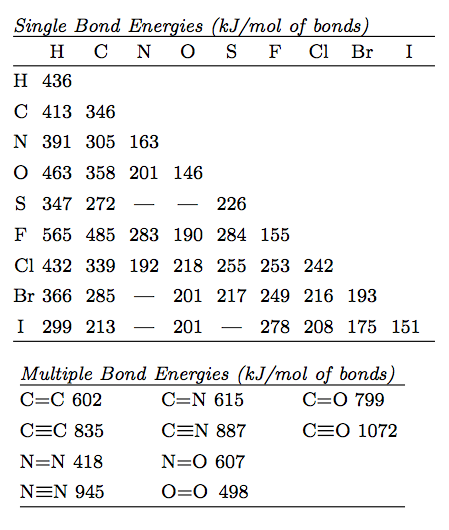
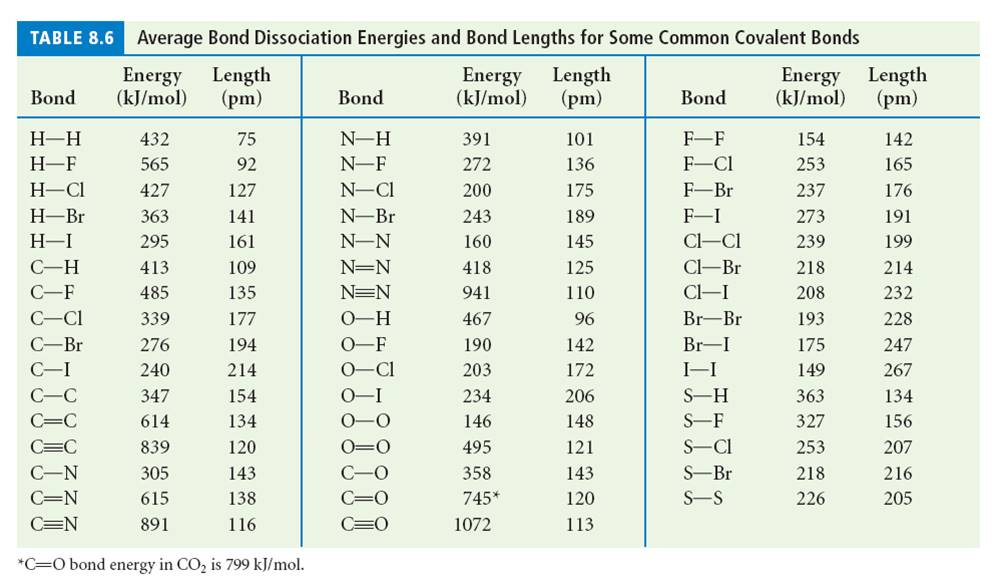
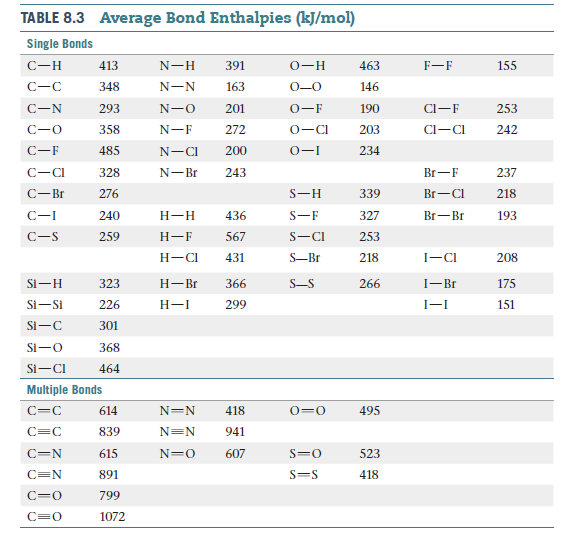
SOLVED: Table 1: Average Bond Energies (kJ/mol) Bond Energy H-H 432 F-F 154 H-F 565 O-H 467 C-H 413 Cl-Cl 239 C-O 358 C-C 614 CO 1072 C-O 745 C-C 347 C-O (for CO2(g)) 799 C-Cl 339 O-O 495
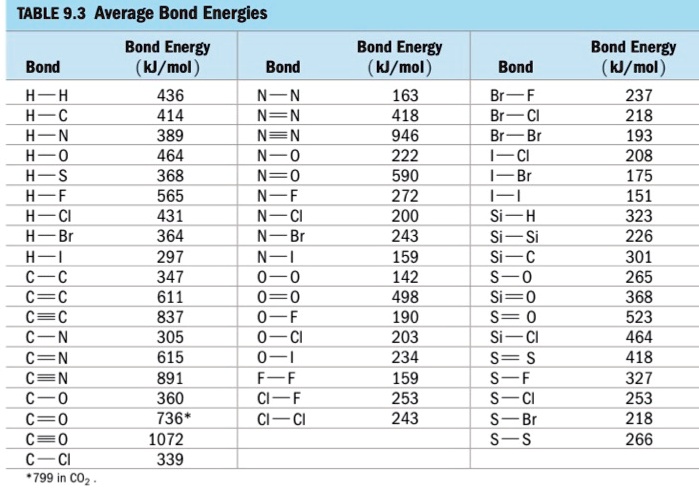
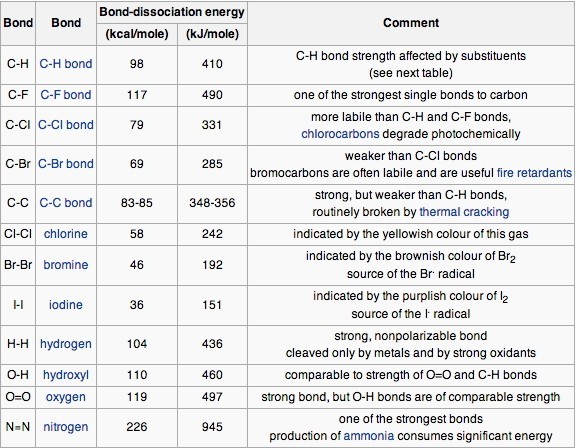
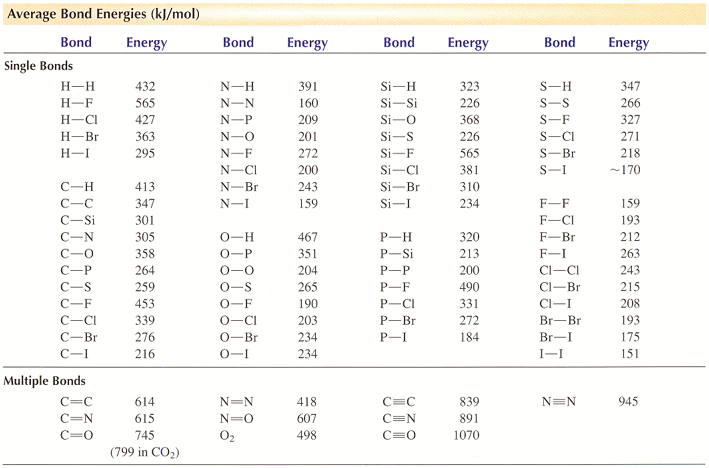
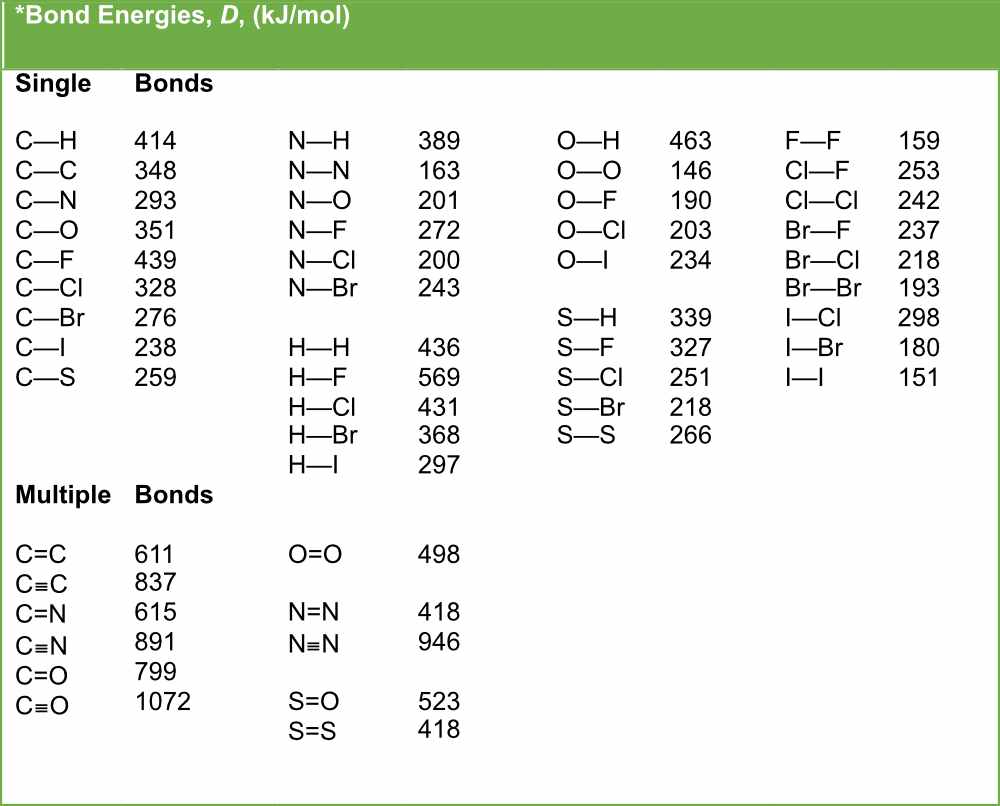
SOLVED: TABLE 9.3 Average Bond Energies Bond Energy (kJ/mol) Bond 436 Bond Energy (kJ/mol) 163 418 946 222 590 272 200 243 159 142 498 190 203 234 159 253 243 Bond Energy (kJ/mol) 237 218 193 208 175 151 323 226 301 265 368 523 464 418 327 253 218 266 ...
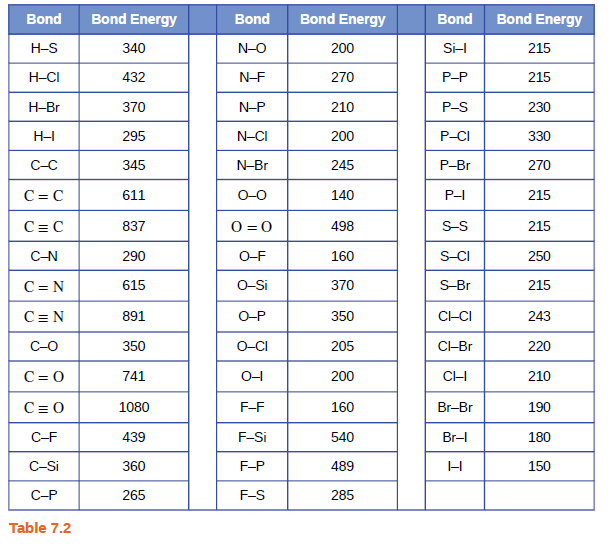
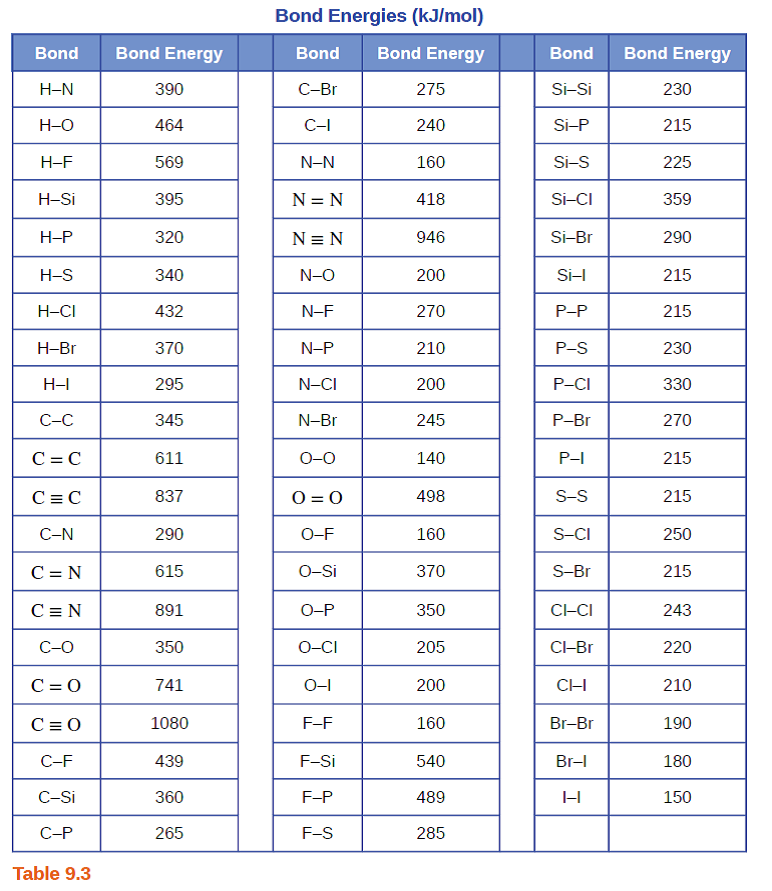
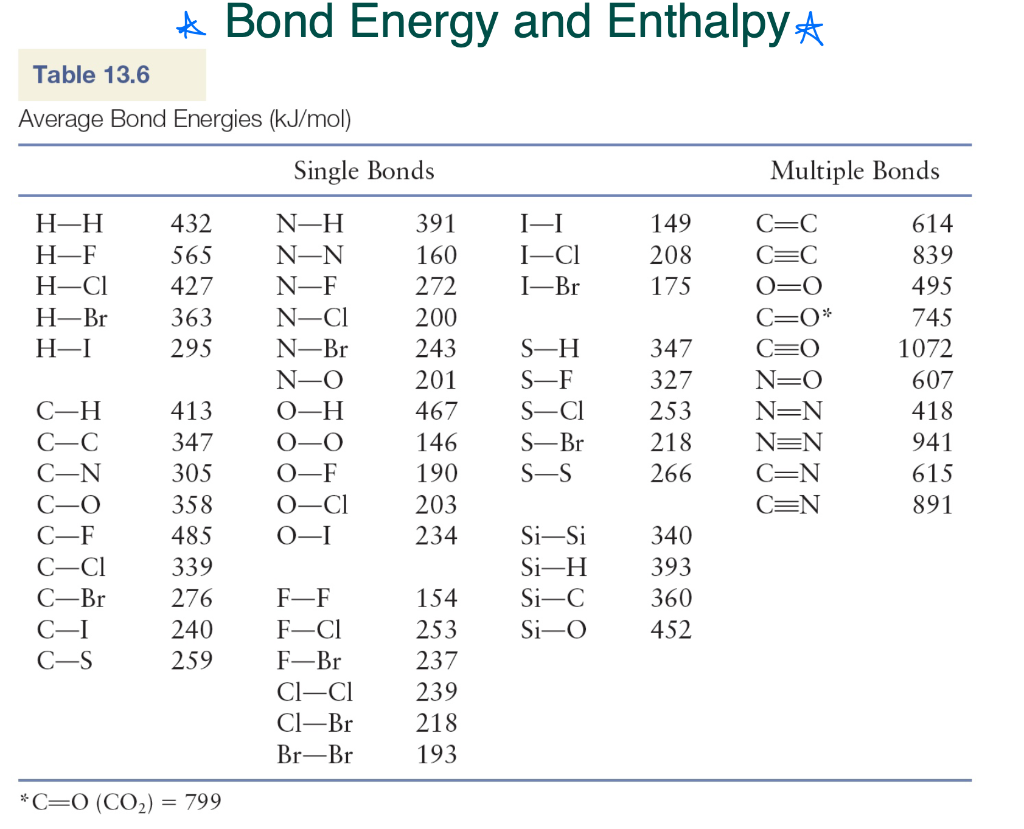
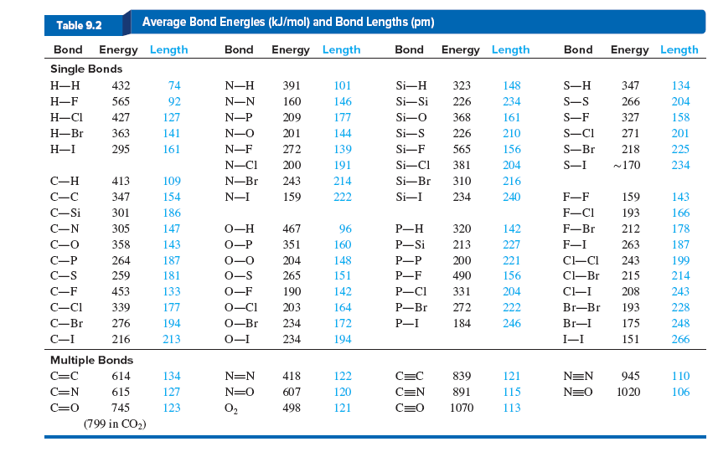
Using the bond energies in Table 7.2, determine the approximate enthalpy change for each of the following reactions: (a) Cl 2 ( g ) + 3 F 2 ( g ) →
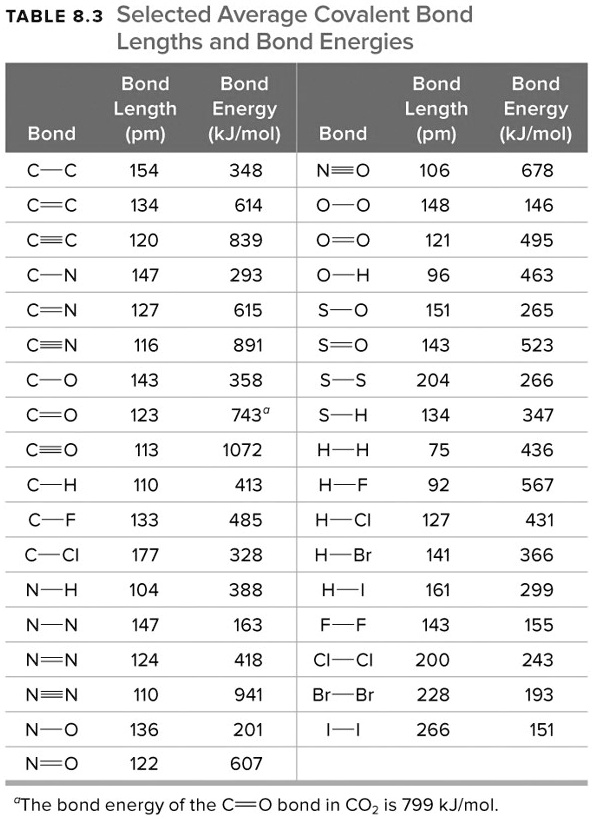


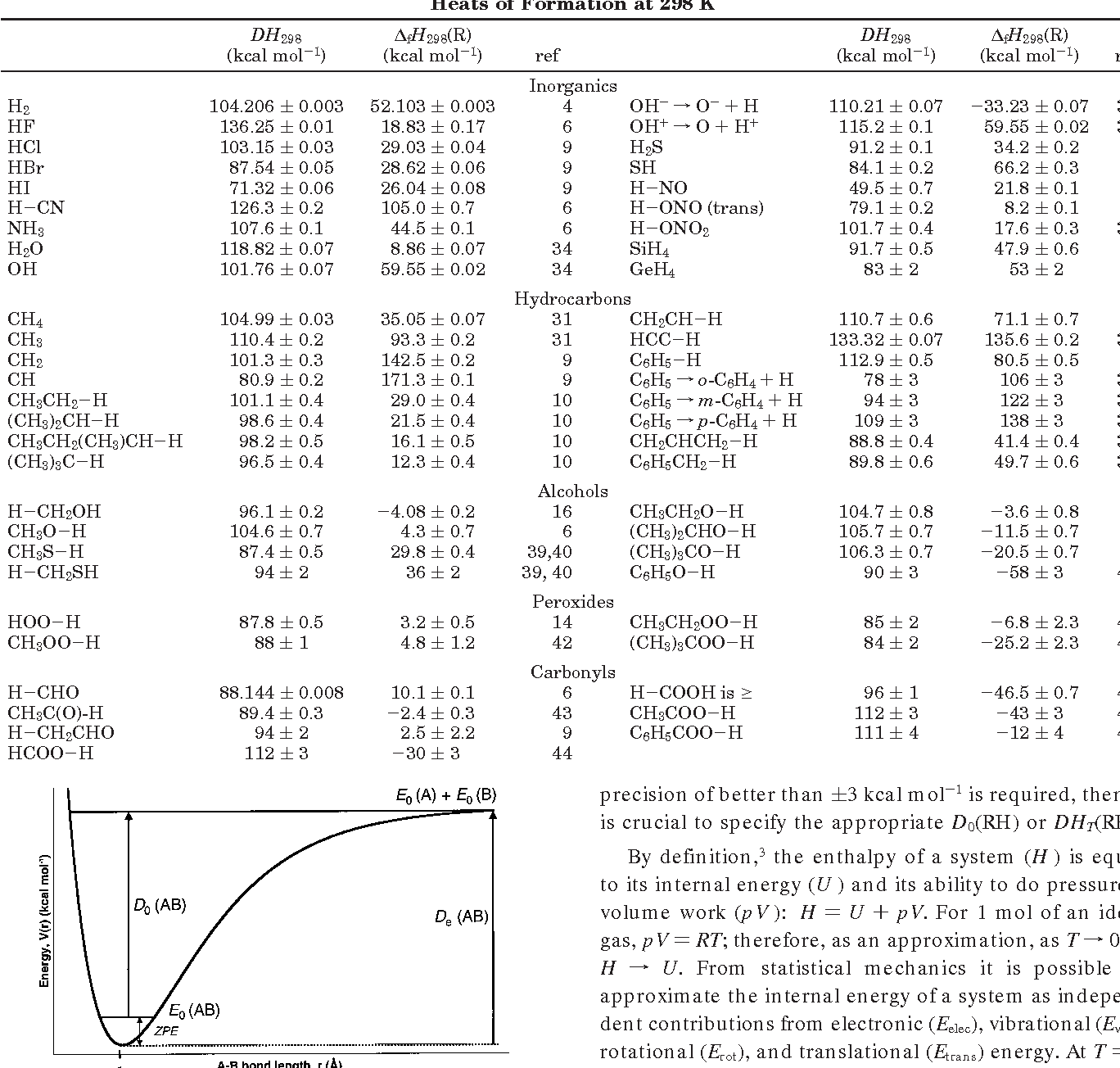
![PDF] Bond dissociation energies | Semantic Scholar PDF] Bond dissociation energies | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/ce7c065d005590afbab6bb30de466e75e50d4f86/7-Table3-1.png)